It is really going to happen! We talked about this for YEARS, and now we are finally going to see it come to fruition.

Kate Loudon and I have known each other for a long time. It was kind of inevitable that two women who were part of the leadership of the Entomological Society of America’s Section B, now the Physiology Biochemistry and Toxicology (PBT) section, would become friends. We are actually from THAT era where female leadership in the ESA was a rarity (not anymore!).
Since we both have an interest in insect physiology (broadly) and biomechanics (specifically) we started talking about organizing a bioinspiration symposium. Fundamental insect biomechanics studies have inspired technologies for some time now. For about 5 years I have been teaching courses on bioinspiration and I use Kate’s research on bed bug-killing materials as an example of innovations that can be inspired by nature and benefit society. So the match seemed natural. Also, we really like each other and would use any excuse to collaborate on something.
It took a while but we managed to put together an awesome symposium with prestigious speakers on the biggest entomological stage ever; the 2016 XXV International Congress of Entomology to be held in Orlando, FL (Sept 25th-30th).
We are so thrilled about the line-up. There is a great variety of speakers (topics, nationality, ethnicity, gender) and we can’t wait for them to interact with each other and other interested entomologists. Some of our speakers have never been to an entomological meeting. We expect to get them hooked, or at least speak well of us entomologists once they are back at their home institutions.
We hope that as a result of this symposium new collaborations will develop, be it to delve into new research questions or to explore educational avenues.
Let me first introduce you to the speakers. Hopefully as the symposium draws closer I can share a little bit more about the topics and speakers in follow-up posts.
- Our first speaker will be Dr. Robert Wood who is the Charles River Professor of Engineering and Applied Sciences in the Harvard John A. Paulson School of Engineering and Applied Sciences. Prof. Wood is also a founding core faculty member of the Wyss Institute for Biologically Inspired Engineering, a power-house in the field of bioinspiration. The Wood lab is probably most famous among entomologists for their work on robobee – a miniature flying, and now also sensing, robot inspired by biology.
- The next speaker will be Chen Li from Johns Hopkins University. Prof. Li coined his own research topic: terradynamics. Similar to how aero- and hydrodynamic principles have shaped our knowledge about animal locomotion in air and water it is Prof Li’s goal to better understand animal locomotion on complex (always shifting) terrains, thus his creation of the terradymics lab at JHU. Cockroaches feature prominently in his research.
- Next we switch from robotics to bioinspired materials. Kate (Dr. Catherine Loudon, University of California at Irvine) will share her work on how small structures on bean leaves kill bed bugs and how these structures (and their special characteristics) have spurred interest in the development of physical insecticidal bioinspired materials.
- Faithful readers of this blog will know by now how enamored I am by the insect cuticle. I am therefore glad that we will have Dr. Stevin Gehrke (Fred Kurata Memorial Professor of Chemical Engineering at the University of Kansas) talk about the physical properties of beetle elytral cuticle and why this type of biomaterial may have many possible applications.

Tenebrio molitor with characteristic elytra covering the hind wings. By gbohne from Berlin, Germany
- Next I will discuss a relatively new project from my lab at the University of Illinois at Urbana-Champaign in collaboration with Dr. Nenad Miljkovic from Illinois’s Mechanical Science and Engineering Department and Dr. Donald Cropek who is a chemist from the U.S. Army Corps of Engineers. Over the past few months we have initiated a comparative study of native Illinois cicadas’s wings to determine physical and chemical attributes that make the wings have (super)hydrophobic, self-cleaning and maybe even antimicrobial characteristics. This collaboration has been really fun and I have learned about a lot of new techniques and I hope to share some of my excitement with the symposium’s audience (Bouncing water droplets anyone?!!!!).

Neotibicen dorsatus at Loda Prairie, July 2016. By Marianne Alleyne
- Another interesting biological material is, of course, spider silk. Dr. Crystal Chaw from Dr. Cheryl Hayashi‘s lab at the University of California at Riverside will explain how studies on the evolution of spider silk have helped in the engineering of artificial silk production.
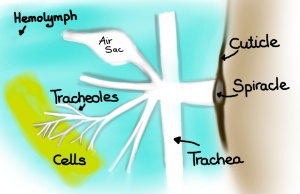
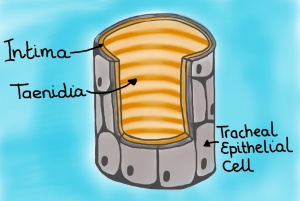
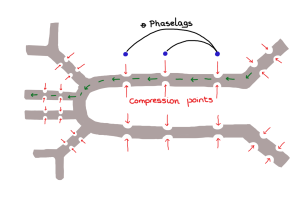
- From biological materials we next move to different types of flow in insects. First Hodjat Pendar from Prof Jake Socha’s lab (Virginia Tech) will be talking about how the insect’s tracheal system (which is actually linked to other physiological systems) can serve as inspiration for novel flow control. It is a fascinating topic which I have touched upon previously in a blog post.
- Flow sensing at a small scale is definitely a topic that is of interest to engineers. And it is something that insects do very well. Dr. Jérôme Casas from the University of Tours (France) will present some of the work he has been doing with Dr. Gijs Krijnen (University of Twente, The Netherlands) on the fluid dynamics of olfaction in insects.
- Another amazing sensor found in insects is the IR sensor in pyrophilous beetles such as in the genus Melanophila. Will we ever be able to engineer an IR sensor as sophisticated as the ones found in beetles? Dr. Helmut Schmitz (Institute of Zoology of the University of Bonn) will share his work to explain how an understanding of the active amplification mechanism seen in the beetle’s IR sensor might help bring us closer to a robust and sensitive bioinspired IR sensor.

IR organ of Melanophila acuminata. Schmitz & Bousack (2012) PLoS ONE 7(5): e37627.
- At this point in the symposium we are shifting gears just a little bit to talk about how to actually DO bioinspired design, and how can we best teach our students to come up with successful bioinspired designs. Most people when they first hear about bioinspiration or biomimicry they immediately think this line of thinking makes total sense. Biologists want to contribute and feel even more justified to delve into fundamental biological questions. Engineers are happy to add bioinspiration into their imaginary toolbox. But for bioinspiration to be successful, to actually have as an end result a bioinspired technology that is based on real biological data, biologists and engineers have to work together. And that is not always so straightforward (Writes the entomologist who has been married to a mechanical engineer for 20+ years. Trust me, it is not straightforward.). Prof. Ashok Goel (Georgia Tech, Co-Director of the Center for Biologically Inspired Design. will discuss some of the cognitive challenges that he has encountered when working with collaborators and students on biologically inspired design projects. What he has learned about how engineers and biologists approach certain problems is fascinating.
- The symposium will again switch topics somewhat by next delving into social insect behavior. First up will be Dr. Ted Pavlic (Arizona State University, Associate Director for Research at The Biomimicry Center at ASU) who will talk about how social insects make group decisions and how that knowledge can be transferred to create smart and adaptive teams of robots.

Eciton hamatum workers on the trail, Jatun Sacha reserve, Napo Ecuador. Alexander Wild.
- We will end the symposium with another social insect talk, this one by Dr. Deborah Gordon (Stanford University). Prof. Gordon will talk about her research on collective behavior in ants and how they have influenced engineered networks.
Kate and I hope you can join us for our symposium, either in person or virtually via Twitter or Instagram (we will use hashtag #ICE2016BioI and #ICE2016) and follow-up blog posts. Feel free to use Twitter to ask questions of the speakers (@Cotesia1).
“See” you on the 29th!