I traveled extensively over the summer; to Austin, TX, all through Western Europe and back to Illinois. All the while I was working on this blog post about bioinspired air-conditioning, which was appropriate because everywhere I went I seemed to have to suffer through heat-wave after heat-wave.
While wishing for Europe to have more air-conditioning units (especially in class-rooms and lecture halls), one of course wonders if that would only exacerbate the problem and make summers even hotter. Progress is constantly being made on making air-condition units more compact, more energy efficient, and thus more environmentally friendly. Inspiration on how to accomplish this has been already been found in natural systems.
Recently Brian Clark Howard wrote an interesting and popular article for National Geographic entitled: “5 Natural Air-Conditioning Designs Inspired by Nature”. Arthropods (termites & ticks) were prominently represented on this list. However, I would like to provide a little bit more detail and corrections to the NG’s list.
The insect examples touched upon in the NG article is that of the termite mound. The most famous architectural example of biomimicry or bioinspiration is the Eastgate Centre Building in Harare, Zimbabwe, which opened in 1996. Architect Mick Pearce and the firm Arup were supposedly partly inspired to build a building suitable for a tropical climate by considering the locally present termite mounds, and build their vision using locally available materials.

Eastgate Centre (with chimneys on roof) in Harare Zimbabwe. (Source: wikipedia.org. Unknown photographer)
Anyone who is even the slightest bit interested in biomimicry knows of the Eastgate Centre, but since the political and economic situation in Zimbabwe has probably only deteriorated since 1996 I was wondering if it was actually still standing and still used as the commercial center it had been envisioned as. So I took my questions to Twitter: [View the story “EastGate Building Harare, Zimbabwe” on Storify]. Turns out that the Eastgate building is still used and stands out prominently in the heart of Harare (and as @ardeans pointed out the building is located right on Robert Mugabe Road, sigh). One of the occupants is actually the United States Embassy.
It is just kind of too bad that the building is based on incorrect biology. Or is it?
First a little bit of background about the inspirational insect. Macrotermes termites (Macrotermitinae) occur over tropical Africa and Asia. There are about 330 species in this genus of relatively large termites. Most of the species build elaborate mounds. The tallest mounds occur in Africa (max of 30 feet, 9 meters). Macrotermes termites cultivate fungi, and spend most of their time, somewhere deep within the mound. The Macrotermes species that has been studied the most is the African species M. michaelseni.

Flickr user Potjie uploaded this picture of a termite mound in Northern Namibia. (http://www.flickr.com/photos/potjie/3408013097/)
Popular wisdom says that in order to optimize fungus growth the interior of the mound needs to be maintained within the narrow range of 29-32°C. To keep keep within this narrow temperature range, despite of fluctuating temperatures during the day- and night time, the large mounds are not just heaps of dirt but in fact incorporate elaborate ventilation holes and air ducts and air pockets, which drive natural ventilation through convection. And remember, these structures are build by a million tiny insects that behave in an organized manner to come up with an architectural masterpiece, every time.
So no wonder that Mick Pearce, born and raised in Southern Africa, was inspired by the the termite mound to design a building in tropical Harare that would have similar features.
Up until recently two models for the termite mound function were proposed and commonly accepted. In case of mounds that were capped a “thermosiphon flow” was created – basically hot air created by the nest rises to the top of the mound where it gets refreshed and is supplied with water vapor through the porous mound walls. This denser air then is forced down below the nest, where the cycle is repeated.
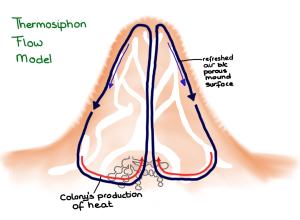
Thermosiphon flow model in which ventilation is driven by heat. (Drawing by Marianne Alleyne, based on Turner & Soar, 2008)
The second model applies to mounds that have a chimney at the top of the mound that opens to the outside. This arrangement creates induced flow, also called the stack effect. The chimney breaks the surface boundary layer and is exposed to higher wind speeds compared to inlets on the ground. The unidirectional flow draws fresh air from near the ground into the nest, where it passes on through the chimney and ultimately to the outside.
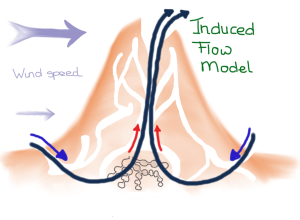
Induced flow model in which ventilation is driven by wind. (Drawing by Marianne Alleyne, based on Turner & Soar, 2008)
The architects who designed the Eastgate Centre building tried to incorporate both the thermosiphon and induced flow principle into their design. The building has an extensive tube system within the walls and floors that move air trough the building. Heat generated within the building, along with stored heat within the structure, creates a thermosiphon-effect that draws air up and through the rooftops where large chimney stacks are located. These tall stacks are essential for creating an induced flow.

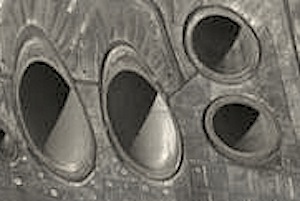
Detail of tube system within Eastgate Centre building’s walls and floors (Original picture at http://harare.usembassy.gov).
When popular stories about the Eastgate Centre building say that the building works on the termite mound principle they ignore the fact that the building uses low capacity fans during the day, and high capacity fans during the night to keep the air from being too stagnant, effectively replacing the hot air that builds up during the day with cool air during the night. This works well, and avoids having to use expensive air-conditioning technology, but needless to say, no termite mound utilizes fans.
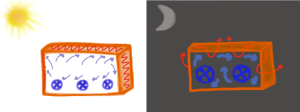
The use of fans within the Eastgate Centre. Smaller fans run during the day-time hours (left). They keep the environment within the building comfortable while the walls store the heat from the outside. Larger fans run during the night-time (right), these fans pull the stored heat out of the walls and push the heat out through the ducting in the ceiling and walls. By the next morning the walls are ready to again store heat (Drawing by Marianne Alleyne).
Since 1996 the common assumptions about how termite mounds are ventilated has been refuted by researchers J. Scott Turner and Rupert C. Soar. And they do it (the disproving) in such a polite manner! (see Conference paper, Flash presentation, YouTube video).
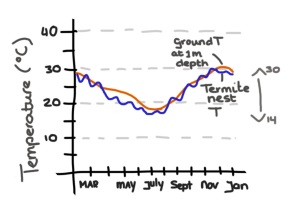
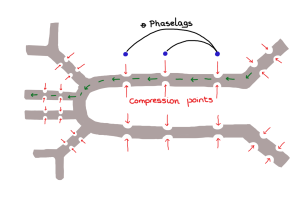
Turner and Soar actually measured temperatures in and outside of the mound. It turns out that while termites may be able to dampen temperatures during the day the nest itself actually closely tracks the soil temperature, which ranges from 15 degrees C in the winter and 31 degrees C in the summer. Mounds have clearly a large thermal capacity, but their architecture, their infrastructure, and their ventilation have little to do with the internal temperature at any given time.
In addition, whereas induced flow might work well in tall buildings because the likelihood of a boundary layer gradient between locations is pretty high, it has been shown that induced flow rarely operates in termite mounds, even open-chimney mounds, since they are commonly only about 6 ft tall. There is also no evidence that mound ventilation and nest ventilation are indeed linked. How respiratory gases are moved from the nest to the mound, and fresh air from the outside, through the mound, to the nest, is not well understood.
Now the Eastgate Centre, and other large buildings since then, accomplished what they set out to do – they saved in construction costs (HVAC systems are very, very expensive) and they save on operating costs, all while keeping the inhabitants comfortable. It probably does not matter that they were based on incorrect science, but it does matter that the misconception gets repeated over and over again. If biomimicry and/or bioinspiration want to be considered legitimate fields of study, and not just a feel-good endeavor, then the science that the field is based on has to be solid.
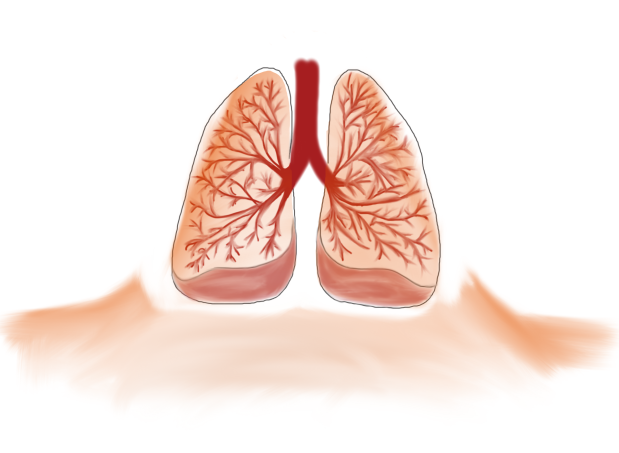
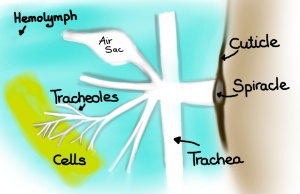
The termite mound should still inspire developers and architects because at it turns out, if we view the termite mound as the analogue of our own, respiratory system (lungs) then we still should be able to design “breathing” buildings that have that walls serve more as membranes rather than barriers.
Turner and Soar not only took relatively simple measurements of the temperatures, they also made plaster casts of the tunnel network of M. michaelseni mounds. They then created horizontal slices of the plaster filled mounds for easier recreation of future 3D models.
Based on these models, which showed actually very little continuous mixing between the air coming down from the above-ground structure and the air from the underground nest, they propose that the termite mound of M. michaelseni is in fact a functional analogue of a lung. And much like the lung the termite mound is far more complex than the simple models we have been using (for an excellent explanation comparing functional organization of both lungs and termite mounds please refer to Turner and Soar, 2008).
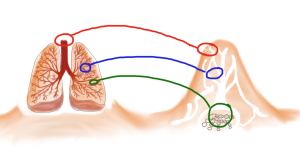
Comparison of the functional organization of mammalian lungs and the termite mound. There are areas of forced convection (large tidal flows) (in red), areas where smaller tidal flows dominate creating a mix of diffusion-convection (in blue), and then the areas where diffusion dominates (in green). (Drawing by Marianne Alleyne, based on Turner & Soar, 2008)
This complexity opens up new avenues for bioinspired design based on the termite mound. Termites erect walls that are actually interface directly with the outside and indoor environments – the walls themselves let gases and energy through, it does not form an impenetrable barrier. Buildings that are designed based on correct termite mound architecture should incorporate porous walls and cladding (~skin) systems that incorporate tubes/tunnels through which air and energy can flow. Maybe we can design living buildings that are part of our extended physiology, as much as the termite mound is part of the living-system called the termite colony.
Resources:
I cannot recommend the Turner and Soar paper from 2008 enough. It is a wonderful read. The authors touch on more topics than I mention here (homeostasis, for instance). And again, their debunking of previously held beliefs is done in a way that should be emulated.
- J. Scott Turner and Rupert C. Soar. 2008. Beyond biomimicry. What termites can tell us about realizing the living building.
- The same authors also wrote a book chapter on a similar topic. But I have not yet been able to locate: Beyond biomimicry. What termites can tell us about realizing the living building. Chapter 15 in: Industrialised, Integrated, Intelligent Sustainable Construction. ISBN 978-0-86022-698-7. Ian Wallis, Lesya Bilan, Mike Smith & Abdul Samad Kazi (eds). I3CON/BSRIA. London. pp 233-248.
J. Scott Turner has also converted this work into very informative video lectures, such as:
- Learning from nature: Termite mound lungs and the implications for breathing mines.
For great footage of how the termite mound models were created check out these videos.
I am not, by a long shot, the first to point out how the termite mound is NOT (yet) the perfect poster-child for bioinspiration and biomimicry. Here are two other blog-posts that discuss this very topic:
- Carl Hastrich on his Bouncing Ideas Blog asked “How is the Eastgate Building not like a termite mound” and if this type of research will encourage us to dive deeper into biomimicry.
- David Parr on his Biomimicron Blog uses the term Biomythology to describe the Eastgate Centre Building‘s place within the biomimicry arena.
Additional pictures of the Eastgate Centre building in Harare Zimbabwe, click here.
Note: I would like to thank Adrian Smith who a few years ago made me aware of the work by Turner and Soar.